Calculating absolute entropy2/6/2024  
For this case, the probability of each microstate of the system is equal, so it was equivalent. Boltzmann's paradigm was an ideal gas of N identical particles, of which Ni are in the i -th microscopic condition (range) of position and momentum. Finally, substances with strong hydrogen bonds have lower values of S°, which reflects a more ordered structure. Interpreted in this way, Boltzmann's formula is the most basic formula for the thermodynamic entropy. For example, compare the S° values for CH 3OH(l) and CH 3CH 2OH(l). Similarly, the absolute entropy of a substance tends to increase with increasing molecular complexity because the number of available microstates increases with molecular complexity. First, calculate the Sreaction based on absolute entropies of reactants and products. Soft crystalline substances and those with larger atoms tend to have higher entropies because of increased molecular motion and disorder. In contrast, graphite, the softer, less rigid allotrope of carbon, has a higher S° due to more disorder in the crystal. Among crystalline materials, those with the lowest entropies tend to be rigid crystals composed of small atoms linked by strong, highly directional bonds, such as diamond. \(\Delta S_\).\( \newcommand\) also reveals that substances with similar molecular structures tend to have similar S° values. If the happening process is at a constant temperature then entropy will be Since there is no disorder in this state, the entropy can be defined as zero. Imagine cooling the substance to absolute zero and forming a perfect crystal (no holes, all the atoms in their exact place in the crystal lattice). Furthermore, it includes the entropy of the system and the entropy of the surroundings.īesides, there are many equations to calculate entropy:ġ. The absolute entropy of any substance can be calculated using equation (1) in the following way. 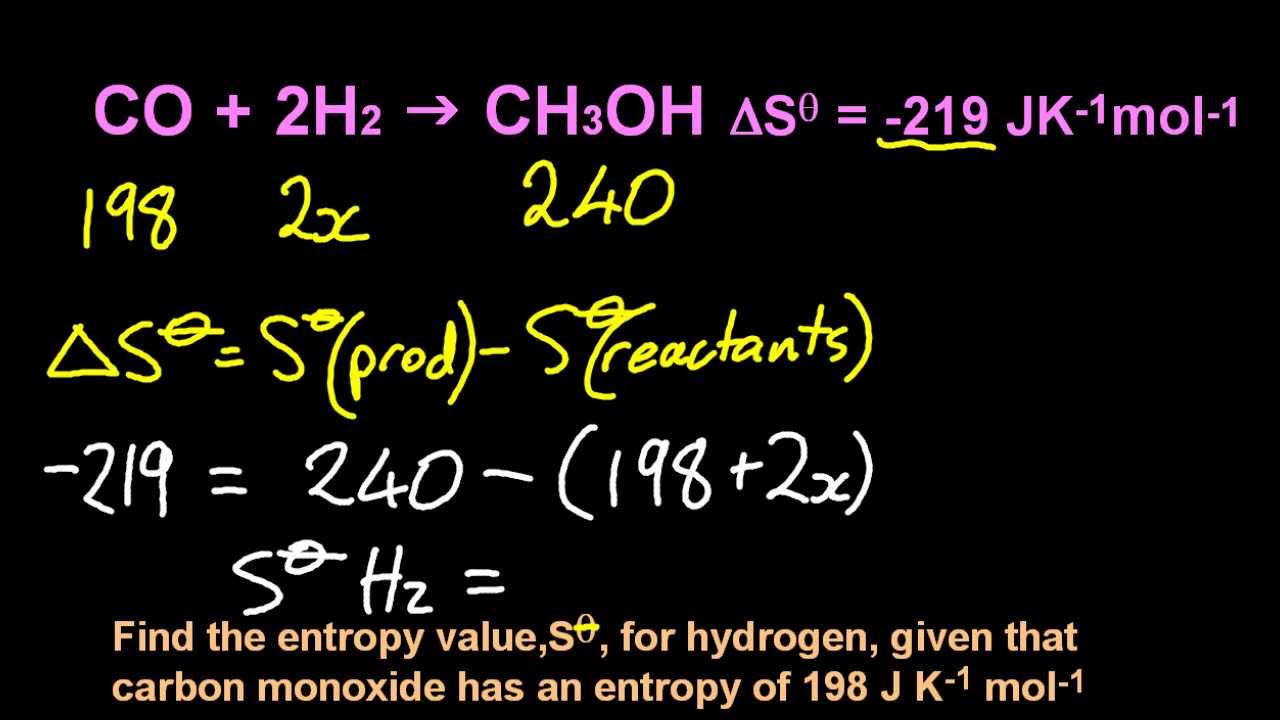
The crystal must be perfect, or else there will be some inherent disorder. Third law: The entropy of a perfect crystal is zero when the temperature of the crystal is equal to absolute zero (0 K). Also, scientists have concluded that in a spontaneous process the entropy of process must increase. The third law of thermodynamics defines absolute zero on the entropy scale. In summary, The concept of absolute entropy is used to set an arbitrary scale for measuring entropy and is based on the third law of thermodynamics which states that the entropy of a perfect crystal at absolute zero is zero. Moreover, the entropy of solid (particle are closely packed) is more in comparison to the gas (particles are free to move). Absolute Definition Entropy Measurement Practical. Entropy FormulaĮntropy is a thermodynamic function that we use to measure uncertainty or disorder of a system. In addition, some microscope process is reversible. Besides, some other example of changeable phase is the melting of metals. On the other hand, blowing a building, frying an egg is an unalterable change. Moreover, when the process is unalterable then the entropy will increase.įor example, watching a movie is a changeable process because you can watch the movie from backward. Also, even when the cyclic process is changeable then the entropy will not change. The second law of thermodynamics says that every process involves a cycle and the entropy of the system will either stay the same or increase. Get the huge list of Physics Formulas here The Second Law of Thermodynamics Furthermore, the more you increase the ball the more ways it can be arranged. So, now you can arrange the balls in two ways. After some time you put another ball on the table. Moreover, the question here is in how many ways you can arrange this ball? The answer is one. In another example, you grab a ball and put it on a table. So, what will happen next? We all know that the smell will spread in the entire room and the perfume molecule will eventually fill the room. Suppose you sprayed perfume in one corner of the room. Furthermore, we can understand it more easily with the help of an example. Moreover, the higher the entropy the more disordered the system will become. Furthermore, you will inspect the formula for entropy and find out how to use it in a variety of cases.Įntropy refers to the number of ways in which a system can be arranged. Moreover, you will explore the second law of the thermodynamics where entropy is introduced. Also, in this topic, we will learn about entropy, entropy formula, its derivation and solved example. Entropy is not a very familiar topic to most of the people. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
